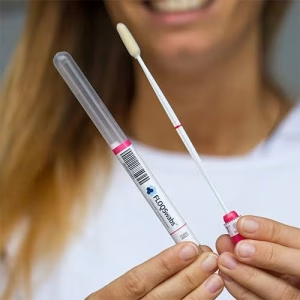
Optimized for Molecular Assays
The eNAT® System is intended to collect, transport, and preserve microbial and human nucleic acids to be analyzed by amplification techniques. With its microbial inactivation and nucleic acid stabilization features, eNAT® is the ready-to-use device to quickly homogenizes samples, for a high-quality nucleic acid yield, for a fast turnaround time.
eNAT® Specimen Collection and Transport Device Optimized for Molecular Assays:
- Stabilizes nucleotides of viral, bacterial, and human samples
- Lyses cells and inactivates potentially infectious specimens
- Prevents bacterial proliferation and target sequence degradation from nucleases
eNAT® is designed for sample collection, transport and preservation of the following types of samples and techniques:
- Respiratory Sampling
- Gastrointestinal diseases
- STI & HPV Testing
- Genetics & Microbiome
- Pharmacogenomics
For the following downstream applications
- Molecular-based assays
- Next Generation Sequencing
*eNAT® is FDA cleared Flu A RNA stabilization with implied use for COVID samples.
Features

High-Quality Nucleic Acid Collection and Transport
eNat® media, a Guanidine thiocyanate-based formula, lyses microbial cells to release and stabilize DNA and RNA, while preventing bacterial proliferation and target sequence degradation from inactivation nucleases.

Securely Handle Infectious Specimens
Securely handle specimens that may contain highly infectious agents, such as COVID-19 using eNAT™ medium which contains a detergent and a protein denaturant to completely inactivate microbial viability within minutes.

Validated and FDA-Cleared
eNat™ has been FDA cleared for Flu A RNA stabilization with implied use for COVID-19 specimens. The system is used with numerous molecular assays for predictive genetics, pharmacogenomics, HLA typing, and microbiome analysis.

Powered by FLOQSwabs® Technology
Expand testing capabilities using eNAT® paired with Copan FLOQSwabs® flocked swabs. Ensure a quick, capillarity-driven sample uptake and a superior elution of more than 90% of the biological specimen, expanding downstream diagnostic testing capabilities.