Genuity Science, a data and analytics organization specializing in high-quality sequencing and advanced software for large datasets and trio analyses for rare disease diagnosis, faced challenges with high sample throughput and complex manual workflows. To overcome these challenges, Genuity Science implemented six Hamilton platforms to automate their NGS workflows from DNA quality control to library pooling. This automation offers significant benefits, including increased throughput, enhanced precision, and improved consistency, while reducing manual errors and integrating seamlessly with Laboratory Information Management Systems (LIMS) for effective sample tracking.
Genuity Science’s Rare Disease Program
Genuity Science, a global life sciences company headquartered in Boston with additional sites in Dublin and Reykjavik, focuses on researching the human genome to explore genetics’ role in health and disease. They collaborate with commercial and academic partners to expand their genomics and health data. Dr. Aoife Coughlan, Program Manager for the Rare Disease Program, aims to use these insights to develop new diagnostics and treatments.
Established in 2017 with Children’s Health Ireland, the Rare Disease Program offers free sequencing services to children with undiagnosed disorders. Analyzing pseudonymized data, the team at Genuity Science works with consultants to confirm diagnoses, with around 20% of the 230 enrolled families receiving a diagnosis. The program continues to re-evaluate cases for new research findings.
The program uses Whole Genome Sequencing (WGS) for comprehensive genetic analysis, alongside Sanger, long-read, and RNA sequencing for confirmation. WGS is preferred for detecting de novo and inherited variants, which is crucial for studying rare diseases.
| Method | Library/Technology | Automated in Hamilton platforms | Purpose |
| Whole Genome Sequencing | Illumina TruSeq PCR free library prep | Yes | Exploratory |
| Whole Exome Sequencing | Agilent’s SureSelect Human All Exon V6 | Yes | Confirmatory; for Copy Number Variation (CNV) analysis |
| Whole Genome Sequencing | Illumina TruSeq Stranded Total RNA library prep | Yes | Confirmatory; to ascertain the impact of novel splice or loss-of- function variants on the canonical transcript |
| Sanger Sequencing | Applied Biosystems BigDye Direct Cycle Sequencing Kit | No | Confirmatory; for analysis of single nucleotide variants |
| Long-Read Sequencing | Oxford Nanopore Ligation Sequencing Kit SQK-LSK109 | No | Confirmatory; for analysis of larger structural variations |
From Manual to Automated Workflows
To handle the increasing sample throughput and complexity of sequencing workflows, Genuity Science sought automated solutions. Dr. George Penman, Principal Scientist of Automation, explains, “We aimed to boost throughput and standardize workflows while integrating sample tracking with our Laboratory Information Management System (LIMS). Manual processes and Excel/.CSV data management became unmanageable as sample numbers grew.”
Genuity Science chose Hamilton for automation due to their systems’ ease of use and flexibility, along with positive references and staff familiarity. Prior to the switch, the company conducted an accreditation and validation process comparing manual and automated workflows. Results from three independent runs showed that both methods provided similar precision (within 5%) and reproducibility (within 2% stdev.), validating the automated approach.
Automated Workflow for Trio Analysis in Patients with Rare Diseases
Two EDTA blood tubes for DNA analysis and one Qiagen PAXgene RNA tube from each family member: father, mother, and proband. DNA and RNA are extracted using an Autogen FlexSTAR Plus and Qiagen QIAsymphony, respectively. Extracted nucleic acids undergo QC, including quantification with Picogreen/Ribogreen, purity assessment with OD 260/280 on a Thermo Fisher Varioskan™ LUX, and integrity evaluation with capillary electrophoresis on an Agilent TapeStation 4200. Hamilton Microlab® STAR™ assists in this process. Passable samples move to the Pre-PCR lab for NGS library preparation on Hamilton STAR™, with workflows automated for Illumina TruSeq and Agilent SureSelect libraries. After library prep, samples are quantified by qPCR with Roche kits, normalized, pooled, and denatured on Hamilton STAR™, then sequenced on Illumina NovaSeq 6000 systems. DNA samples also undergo genotyping on an Illumina iScan system as part of the QC process.

Figure 1: Complete workflow of the analyses performed on samples from patients on the Rare Disease program. Steps shown in blue are performed on Hamilton platforms
Description of the Hamilton Systems
Genuity Science utilizes six Hamilton Microlab® STARs™ across three labs: two in the isolation lab, two in the Pre-PCR lab, and two in the Post-PCR/Sequencing lab. Each STAR™ is equipped with eight 1000 μL CO-RE® channels, a CO-RE® 96 Probe Head, a CO-RE® Gripper, and an Autoload with a 1D Barcode Reader. Models with the Hamilton On-Deck Thermal Cycler (ODTC) also include an iSWAP. Additional tools and modules specific to each platform are depicted in Figure 2.
In the isolation lab, the STARs™ handle the setup of 96 and 384 well plates for DNA QC (quantification, purity, integrity) and NGS library prep. In the Pre-PCR lab, they prepare NGS libraries, and in the Post-PCR/Sequencing lab, they assist with library prep amplification (if needed), setup 364 well plates for qPCR, and normalize and pool libraries into NovaSeq library tubes.
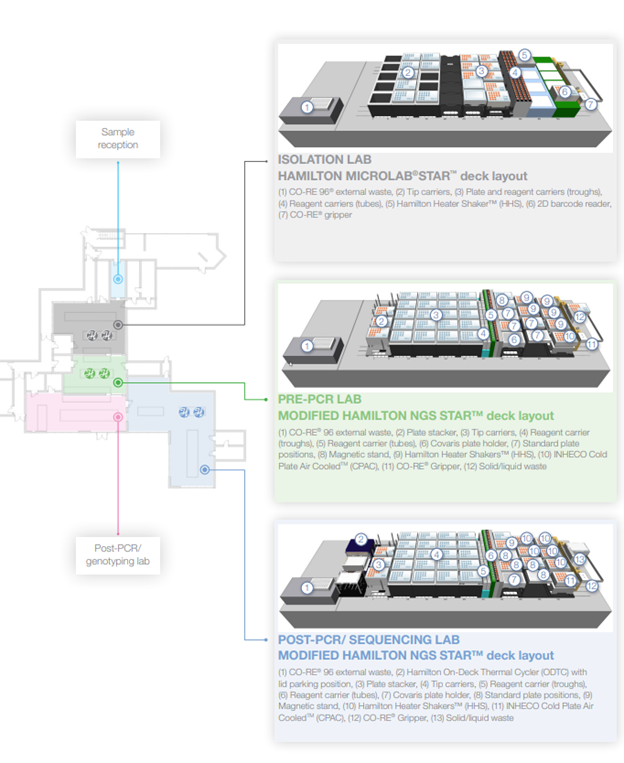
Figure 2: Representative deck layout of the six Hamilton Microlab® STARs™ in the laboratories of Genuity Ireland
The automation of workflows with Hamilton platforms has allowed Genuity Science to customize methods, track samples, and boost throughput:
- Flexibility and Customization: Hamilton’s Assay-Ready Workstations include pre-qualified methods for NGS library prep from Illumina. Genuity Science adapted these methods and created new protocols for NGS library prep and QC using Hamilton’s software.
- Sample Traceability: With numerous samples and steps, Genuity Science implemented rigorous tracking. Hamilton’s on-deck traceability and LIMS integration ensure full sample traceability. Dr. Penman highlights that 2D barcode readers in the isolation lab link gDNA samples to all plates and tubes, including those for the NovaSeq 6000.
- Increased Throughput: Automation has increased Genuity Science’s capacity to 2,000 genotyping runs and 960 WGS library preparations per week using Illumina’s TruSeq PCR-free workflow, benefiting all programs, including the rare disease program.
------------
GENESMART CO., LTD | Phân phối ủy quyền 10X Genomics, Altona, Biosigma, Hamilton, IT-IS (Novacyt), Norgen Biotek, Rainin tại Việt Nam.











